Through ET, a single, genetically superior female is able to generate a greater number of offspring than through conventional breeding systems (natural service or A.I.) and, when coupled with sperm from a genetically outstanding sire, embryos of exceptional genetic quality can be produced. In addition, recipient females of poor or average genetic merit have the opportunity to serve as surrogates and receive an embryo with high genetic value; therefore, these recipients can give birth to calves with greater genetics than they would otherwise.
Through ET, genetic progress can be hastened, which is particularly useful in cattle due to their relatively long generation interval when compared to other livestock species. Another great advantage of this technology is the ability to transport embryos to areas where the production of beef needs to be advanced instead of the live animals themselves.
Currently, there are two methods utilized commercially for embryo production in cattle ET programs: multiple-ovulation ET (MOET) and in vitro fertilization (IVF) followed by ET. In current MOET protocols, donor females are superovulated through treatment with follicle-stimulating hormone (FSH) in order to stimulate the release of multiple oocytes at the time of ovulation. These superovulated females are A.I.'d and, if successful, a number of embryos will be recovered at the time of uterine flushing.
Following embryo collection through the uterine flushing procedure, embryo viability is determined, and viable embryos are either transferred fresh to recipient females or are frozen for future use. On average, 6.9 viable embryos are recovered per flush in beef females; however, this number fluctuates depending on cow breed, age and within-breed variation. The transfer of fresh embryos typically yields 10% to 15% greater pregnancy rates than the use of frozen-thawed embryos. Another important contributor to MOET success is the semen utilized to A.I. donors, as it has been well documented that donors' A.I. with sex-sorted semen produce fewer embryos than donors' A.I. with conventional semen.
Superovulation protocols increase the number of follicles ovulated per estrous cycle, allow multiple oocytes to be fertilized at a time and ultimately lead to the production of several embryos at once. Currently, the recommended superstimulatory protocol for Bos taurus donors involves inserting a CIDR on day zero, followed by a 100-μg injection of GnRH two days later.
Beginning on day four, donors receive injections of FSH every 12 hours. The amount of FSH per injection decreases each day until day seven, resulting in a total of eight injections and 400 milligrams of FSH. On day seven, donors receive two injections of prostaglandin F2α (PGF2α) 12 hours apart (a.m./p.m.). At the time of the second PGF2α injection, the CIDR insert is removed, and heat detection begins 24 hours after CIDR removal and continues until day 11. Donors detected in heat during this period are A.I.'d at both 12 and 24 hours after onset of estrus.
Embryos are flushed seven days after A.I. (Figure 1).

A major limitation to the production of embryos from MOET has been the lack of reliability at successfully inducing superovulation in donor females. However, research into the development of superovulation protocols and techniques to predict which donor females may respond well to superovulation is ongoing. Superovulation can be performed by the producer without the need for a specialist; however, superovulatory drugs are expensive, and donors need to be well managed.
An alternative to embryos derived from MOET is the production of embryos in a laboratory via in vitro maturation, IVF, and in vitro embryo culture, which are collectively referred to as IVF. IVF can generate pregnancies from a donor female that is already pregnant and requires fewer units of semen when compared to MOET. In fact, one single semen straw can provide enough sperm to fertilize as much as 800 to 1,000 oocytes.
Furthermore, oocytes can be collected from the antral follicles of ovaries obtained from slaughter facilities, which greatly increases the number of embryos that can be produced and can hypothetically eliminate the need for donor females. Finally, the potential disadvantage of a poor response to a superovulation protocol can be avoided by utilizing IVF.
During the IVF process, oocyte maturation needs to take place, where oocytes complete their first meiotic division. Similarly, spermatozoa used for IVF need to undergo capacitation before they are able to penetrate the zona pellucida of the oocyte. Oocytes that mature spontaneously in vitro or in vivo are highly capable of sperm penetration. However, oocytes matured in vitro have shown reduced developmental capacities in comparison to those matured in vivo. In addition, the viability of IVF-derived embryos decreases with cryopreservation to a greater extent than in vivo-derived embryos; therefore, these embryos are more commonly transferred fresh.
The predominant oocyte collection technique is known as aspiration or ovum pickup (OPU). Through OPU, unfertilized oocytes can be harvested directly from the ovaries of a donor cow or heifer using an ultrasound probe and an aspiration needle. This technique may be performed two to three times during a cow’s estrous cycle for as long as six months, which is more frequent than what MOET can be performed. Therefore, a greater number of transferrable embryos per donor can be generated through IVF than through MOET. On the other hand, embryos produced by IVF are known to yield pregnancy rates approximately 10% lower compared to embryos produced by MOET. Furthermore, pregnancy losses throughout pregnancy are greater in IVF embryos compared to MOET.
Both IVF and MOET can be successfully incorporated into beef cattle operations to enhance genetic improvement; however, either technique is not necessarily suitable for all cattle operations. There are a number of considerations that need to be made when selecting an ET technique, such as labor availability to administer hormones during a superovulation protocol, adequate donor management, number of embryos generated and pregnancy rates, as well as the cost-effectiveness of either protocol. Overall, nutrition, estrous cycle control and donor and recipient management all contribute to the success or failure in any given ET program; therefore, producers, embryologists, veterinarians and all members of the herd management team need to work together in order to maximize ET success. ![]()
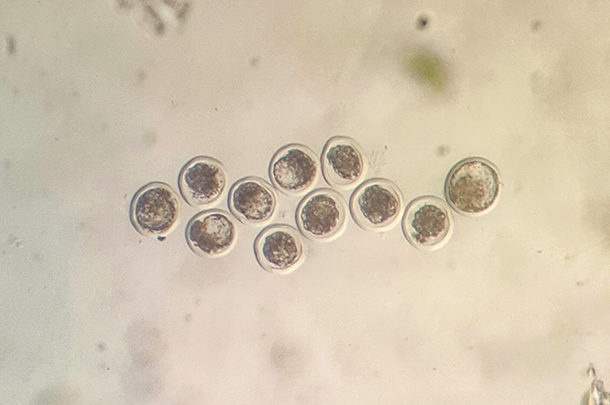
PHOTO: In vivo-produced embryos. Photo by Nicky Oosthuizen.
G. Cliff Lamb is a professor and head of the department of animal science at the Texas A&M College of Agriculture and Life Sciences. Email G. Cliff Lamb.

-
Nicky Oosthuizen
- Ph.D. Student, Graduate Research Assistant
- Texas A&M University
- Email Nicky Oosthuizen